By Narendra Chirmule, PhD, Sr. VP & Head of Research and Development at Biocon.
Biotechnology has the promise to make highly efficacious drugs (biologics) which have minimal side effects utilizing our own biological material.
Let’s start with a story:
Why do we get Anaemia? Anaemia is a condition that develops when your blood lacks enough healthy red blood cells (also called erythrocytes), which transport hemoglobin. Hemoglobin is a part of erythrocytes and binds oxygen. If you have too few or abnormal erythrocytes, or your hemoglobin is abnormal or low, the cells in your body will not get enough oxygen.
How are erythrocytes made by the body? Production of erythrocytes is controlled by Erythro-poietin (EPO), a hormone produced primarily by the kidneys. Erythrocytes start as immature cells in the bone marrow and after approximately seven days of maturation are released into the bloodstream.
So, how would one treat Anaemia? With Erythropoietin, silly!

What is this ….Erythropoietin? Approved by the FDA to treat anemia first in 1989, erythropoietin is the poster child of the field of biotechnology.
Erythropoietin was discovered in 1906 (1). It took another 40 years to discover that EPO played a central role in development of erythrocytes in the bone marrow (2), and another 10 years to identify the source as the kidneys (3). Next, the EPO protein was purified from urine of patients suffering from various diseases (4). The protein is a 34-kilo Dalton glycoprotein hormone containing 193 amino acid peptide sequence. Proteins are expressed from genes. The next landmark was 1985, when the DNA sequence of human EPO gene was determined after three years of intensive research (5).
How is erythropoetin made “outside the body?” Using recombinant DNA technology. The techniques of recombinant DNA simply refers to the transfer of a gene from one organism into another organism: literally, the recombination of DNA from different sources.
Lets follow the process carefully…it is quite simple…
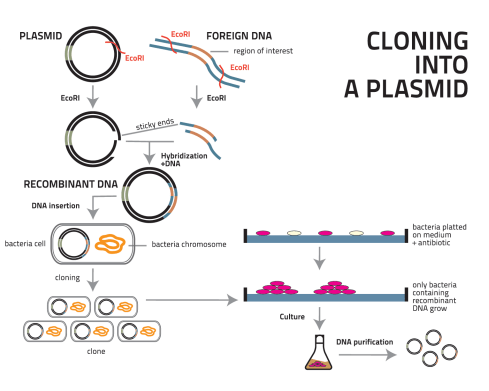
In the case of EPO, the recombinat DNA process involves isolating EPO gene from a kidney cell, and introducing it into bacteria, called Escherichia coli (E. coli).
- Extremely specific enzymes that cut DNA, called restriction enzymes, can excise individual genes from human DNA.
- The excised gene (EPO, in this case) is isolated and inserted into small circular pieces of DNA cut with the same restriction enzyme, known as plasmids.
- Once inserted into a plasmid, the EPO gene can be glued in place using another enzyme called DNA ligase. [Restriction enzymes and DNA ligase are the scissors and glue of recombinant DNA technology.]. Thus far, the EPO gene has been cut from the DNA of the kidney cell, and inserted into the DNA of the bacteria, E.coli. In other words the E.coli are transformed.
- Transformed bacterial cells are separated from non-transformed cells in a selection procedure that takes advantage of drug-resistance genes that is strategically present on the plasmid DNA.
- In the presence of a chemical drug during the growth of bacteria, non-transformed bacteria die, resulting in purification of the transformed bacteria, which contain the EPO gene, and express (secrete) EPO protein, now called recombinant-human EPO (rhEPO).
- A pure population of recombinant transformed bacterial cells is then established through the process of cloning. In cloning, a single cell is selected, and it gives rise to a whole population of identical cells, or clones, by normal cell division. In this process, all of the resultant cells are expected to contain a copy of the plasmid carrying the inserted EPO gene.
- The protein is purified, put into vials and syringes, and is be used to treat patients with anemia.
The first clinical trial using rhEPO in the treatment of the anaemia of end-stage renal failure was published in 1987 [6]. Since then, many therapeutic trials have been developed to demonstrate the potential use of rhEPO in the treatment of different kinds of anaemia, namely associated with prematurity, AIDS, malignancies, congestive heart failure, post chemotherapy and post transplantation [8].
Summary: The use of EPO to treat anaemia is the most natural process of increasing the red blood cells that is the cause of disease. The techniques of recombinant DNA technology used to manufacture EPO in bacteria is biotechnology.
References:
- Carnot, P.; Deflandre, C. Sur l´activite hemopoietique du serum au cours de la regeneration du sang. C. R. Acad. Sci., (Paris) 1906, 14, 384-386.
- Bondsdorff, E.; Jalavisto, E. A humoral mechanism in anoxic erythrocytosis. Acta Physiol. Scand., 1948, 16, 150-170
- Jacobson, L.O.; Goldwasser, E.; Fried, W.; Ptzak, L. Role of the kidney in erythropoiesis. Nature, 1957, 179, 633-634.
- Miyake, T.; Kung, C.K.H.; Goldwasser, E. Purification of human erythropoietin. J. Biol. Chem., 1977, 252, 5558-5564.
- Lin, F.K.; Suggs, S.; Lin, C.H.; Browne, J.K.; Smalling, R.; Egrie, J.C.; Chen, K.K.; Fox, G.M.; Martin, F.; Stabinsky, Z.; Badrawi, S.M.; Lai, P.; Goldwassert, E. Cloning and expression of the human erythropoietin gene. Proc. Natl. Acad. Sci. USA, 1985, 82, 7580-7584
- Eschbach, J.W.; Egrie, J.C.; Downing, M.R.; Browne, J.K.; Adamson, J.W. Correction of the anemia of end-stage renal disease with recombinant human erythropoietin. Results of a combined phase I and II clinical trial. N. Engl. J. Med., 1987, 316, 73-78.
- Stryjewska, Agnieszka et al. “Biotechnology And Genetic Engineering In The New Drug Development. Part I. DNA Technology And Recombinant Proteins”. Pharmacological Reports 65.5 (2013): 1075-1085.