DCGI Approval for emergency use of Itolizumab in treatment of Covid-19
Biocon’s ‘first-in-class’ anti-CD6 monoclonal antibody, a novel biologic, Itolizumab, received the DCGI’s nod for its emergency use in treatment of CRS in moderate to severe ARDS patients due to COVID-19, based on the results of a ‘proof of concept’ pivotal Phase II clinical study.

A multi-centric, open label, two-arm randomized pivotal clinical trial was conducted in 30 eligible patients at four hospitals across Mumbai and New Delhi. Twenty patients received Itolizumab plus best supportive care in the test arm, while 10 patients received best supportive care alone in the control arm. The primary endpoint was mortality at the end of one month.

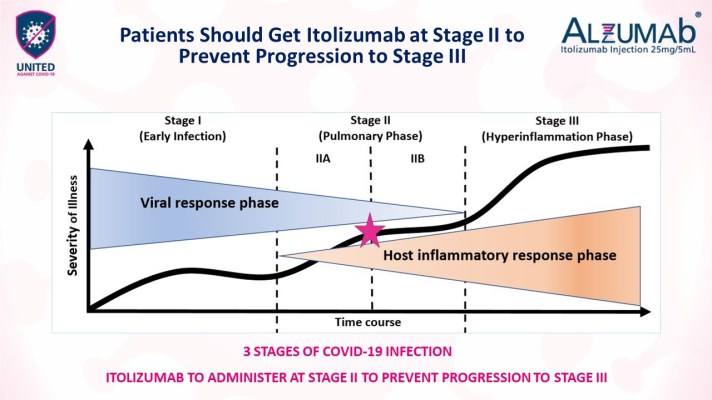
At the end of the treatment period, Itolizumab demonstrated statistically significant advantage over the control arm in one-month mortality rate. All 20 patients on drug arm who were administered Itolizumab recovered fully and were discharged from hospital. Whereas three out of ten patients in the control arm with best standard of care died while seven recovered. Key efficacy parameters of lung function such as PaO2 and SpO2 (oxygen saturation) improvement without increasing FiO2 (oxygen flow) also showed statistically significant advantage for the Itolizumab arm over the control arm. All patients on the Itolizumab arm were weaned off oxygen by Day 30, and none needed ventilator support unlike the control arm.
Key secondary endpoints of clinical markers of inflammation such as IL-6, TNF-α, serum ferritin, d-dimer, LDH and CRP showed clinically significant suppression post Itolizumab dosing and correlated well with clinical improvement in symptoms and chest X-ray images.


Itolizumab was overall well tolerated and found to be safe. The infusion reactions in some patients were manageable with slowing down the infusion rate.
Pan-India Phase IV trial to be conducted across 10-15 hospitals
As a part of our commitment to the DCGI, we have planned a 200-patient, pan-India Phase IV trial to be conducted across 10-15 hospitals that have a high caseload of serious COVID-19 patients. The study protocol has been submitted to the DCGI and we will commence the trial soon.
The results from the Phase IV trial will create a larger body of evidence for efficacy of Itolizumab in COVID-19 complications, which will be included in our research publications.
Expert Panel Discussion
An expert panel discussion was held on the role of Itolizumab in treating COVID-19 patients which was telecasted on Rajya Sabha TV.
Dr. Ram Vishwakarma from CSIR, Dr. Shashank Joshi, Dean Indian College of Physicians, Dr. Sandeep Athalye, Chief Medical Officer, Biocon Biologics were part of the expert panel.
Expert Opinion: Peer Reviewed Publications on Itolizumab
Itolizumab an anti CD6 monoclonal antibody as a potential treatment for COVID 19 complications, a peer-reviewed article published in the Journal, ‘Expert Opinion on Biological Therapy’ authored by Subramanian Loganathan , Sandeep N. Athalye & Shashank R. Joshi

Read the full article here: Itolizumab an anti CD6 monoclonal antibody as a potential treatment for COVID 19 complications
Expert Opinion
Dr. Suresh Kumar, Medical Director, LNH, Delhi, Principal Investigator, Pivotal Phase II, Itolizumab Study talks about the effectiveness of Itolizumab in targeting the COVID-19 virus.
Publications
DCGI has been proactive, responsible and transparent, claims Biocon, an article by Dr. Sundar Ramanan, Global Head of Regulatory Affairs at Biocon Biologics which corrects with evidence the factual inaccuracies of the opinion article titled, “An opaque model of drug approval” published in The Telegraph.
The article was published in The Telegraph on 28th July 2020.

Biocentury
An article published in BioCentury Magazine talks about how Biocon is planning to run larger studies and collect real-world data that can demonstrate the CD6 inhibitor can effectively dampen the cytokine storm observed in severe COVID-19 patients, a feat IL-6 inhibitors have yet to demonstrate.

Read the full article here: Will Biocon’s COVID-19 anti-inflammatory succeed where IL-6 blockers have failed?
Live Mint
Antibody drug lowers risk of death in elderly coronavirus patients: Study – The article talks about Biocon Ltd’s regulatory approval in India & Cuba for Itolizumab for use in coronavirus infected patients. The researchers, who developed Itolizumab, said timely use of the drug in combination with standard therapy helped reduce inflammation and prevented COVID-19 from worsening.

Read the full article here: Antibody drug lowers risk of death in elderly coronavirus patients: Study
Equillium plans to conduct a global randomized controlled clinical trial of Itolizumab
Based on the encouraging study results, our U.S.-based partner, Equilllium is planning to conduct a global randomized controlled clinical trial of Itolizumab in COVID-19 patients for which it will file a U.S. investigational new drug application (IND) soon.
Equillium PR: CLINICAL TRIAL SHOWS ITOLIZUMAB REDUCES MORTALITY IN PATIENTS HOSPITALIZED WITH COVID-19
Cuban regulatory authority approves Itolizumab for the treatment of seriously and critically-ill COVID-19 patients
Our Cuban partner, Centre of Molecular Immunology, has reported encouraging results from a clinical trial with Itolizumab in 76 COVID-19 patients in Cuba. At the end of the trial, 79% of severely ill patients were discharged from the ICU after 14 days of treatment, while moderately ill patients showed a reduction in the rate of disease progression.
Read More: Emergency use of Cuban medicine against COVID-19 approved

